Understanding Your Risk of Cervical Cancer After a LEEP Procedure

Key points
- Negative Margins: This is the ideal outcome. It means there are no abnormal cells at the edges of the removed tissue, suggesting the entire lesion was successfully excised.
- Positive Margins (or Involved Margins): This means abnormal cells were found at the edge of the tissue sample. It indicates that some precancerous cells may have been left behind on the cervix.
If you've been told you need a Loop Electrosurgical Excision Procedure (LEEP), it's natural to have questions and concerns, especially about what comes next. A LEEP is a common and highly effective procedure used to remove abnormal, precancerous cells from the cervix. While its primary goal is to prevent cervical cancer, many wonder about the long-term risk. Navigating abnormal cervical screening results can be emotionally overwhelming, and it is completely normal to experience anxiety about your fertility, future cancer risk, and what daily life will look like during recovery. Understanding the clinical evidence behind LEEP outcomes can help replace uncertainty with actionable knowledge. This comprehensive guide synthesizes data from clinical studies and medical experts to provide a clear picture of the chances of developing cervical cancer after a LEEP, the factors that influence your risk, and the critical importance of follow-up care. Cervical dysplasia is highly treatable, and LEEP remains one of the most effective interventions in modern gynecology. By understanding the procedure, interpreting your pathology results, and committing to long-term surveillance, you can confidently move forward with your reproductive health journey. It is also helpful to recognize that the evolution of cervical screening has dramatically reduced cancer incidence worldwide. The transition from Pap-only screening to primary HPV DNA testing, combined with risk-based management guidelines, means that precancerous lesions are now detected and treated earlier than ever before, fundamentally changing the prognosis for patients undergoing excisional therapy.
The LEEP Procedure: A Powerful Tool for Cervical Cancer Prevention
A LEEP is performed after abnormal cells are detected during a Pap smear or colposcopy. It uses a thin, low-voltage electrified wire loop to precisely cut away the affected tissue from the cervix. This tissue is then sent to a lab for further analysis. The procedure typically takes place in an outpatient clinic or hospital setting and usually requires only 10 to 20 minutes to complete. The procedure serves two purposes:
- Diagnostic: It confirms the severity of the abnormal cells (cervical intraepithelial neoplasia, or CIN).
- Therapeutic: It removes the precancerous cells, which in many cases is the only treatment needed.
Before the procedure begins, your healthcare provider will typically administer a local anesthetic, such as a paracervical block, to numb the cervix and surrounding tissue. This minimizes discomfort to brief cramping or a slight pressure sensation. A speculum is gently inserted to visualize the cervix, and the electrified wire loop is carefully guided over the targeted area. The electrical current simultaneously cuts the tissue and cauterizes blood vessels to minimize bleeding. Once the specimen is collected, it is placed in a preservative solution and dispatched to a pathology laboratory, where technicians examine it under a microscope to grade the lesion and assess surgical margins.
Prior to the excision itself, most clinicians will apply acetic acid (vinegar solution) and sometimes Lugol’s iodine to the cervical surface during a colposcopy to highlight abnormal areas. This visual mapping ensures that the LEEP loop targets the exact zone of transformation where dysplastic cells are most likely to reside. The depth and width of the excision are carefully calibrated based on colposcopic findings, transformation zone type, and patient age. In younger patients or those with visible lesions, a standard single-pass excision is often sufficient. For patients with a deep or endocervical-predominant lesion, a larger or multi-pass technique may be utilized to ensure complete removal while preserving as much healthy stromal tissue as possible. The electrical settings on the LEEP unit are meticulously adjusted to balance cutting efficiency with hemostasis, reducing thermal artifact at the specimen margins. This technical precision is crucial because excessive heat can distort tissue architecture, making it more challenging for the pathologist to evaluate margin status accurately.
LEEP is considered a cornerstone of cervical cancer prevention, with a success rate of over 90% in removing abnormal cells and preventing progression to cancer. Because the procedure preserves the vast majority of healthy cervical tissue, it is highly compatible with future fertility for most patients. However, the physical and psychological aspects of the experience warrant attention. Patients often report feeling relieved once the abnormal tissue is removed, but they may also need time to process the implications of a precancer diagnosis. Open communication with your care team about anesthesia options, pain management strategies, and what to expect during and immediately after the procedure can significantly improve the overall experience. Many clinics now offer guided breathing exercises, calming music, or the option to have a support person present to reduce procedure-related anxiety. Understanding that LEEP is an outpatient, same-day procedure with minimal downtime can also alleviate fears about hospitalization or extended recovery.
Illustration of the LEEP procedure. Source: Cleveland Clinic.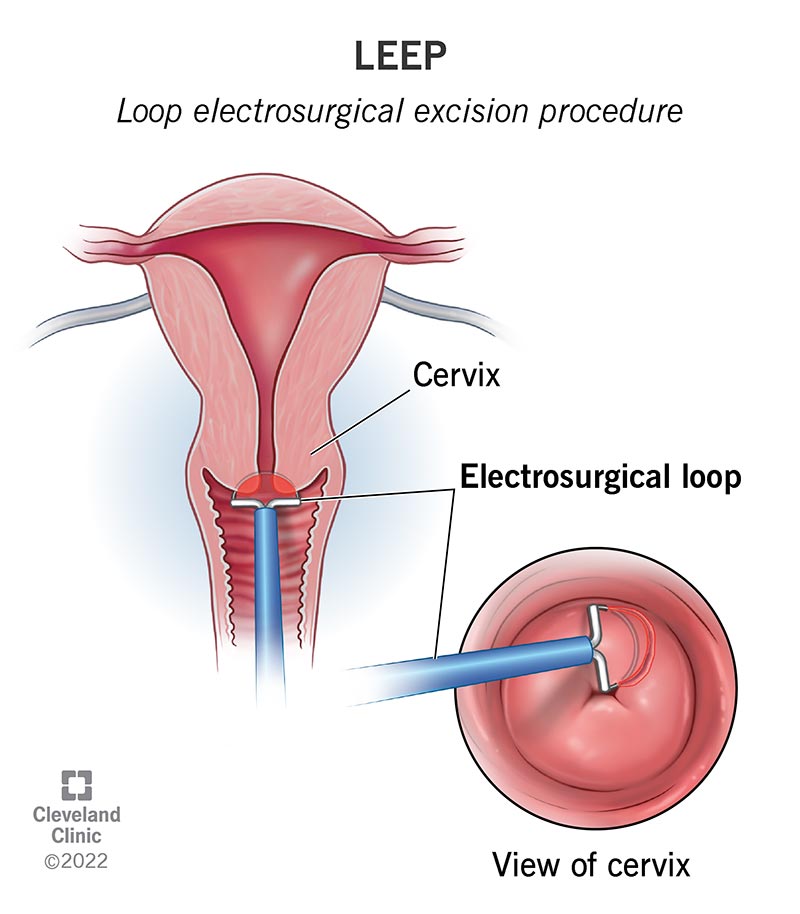
What Are the Chances of Cervical Cancer After LEEP? The Statistics
First, the reassuring news: the risk of developing invasive cervical cancer after a successful LEEP is very low. Medical experts estimate this risk to be around 1-2%. This statistic represents long-term outcomes across diverse patient populations and underscores that the overwhelming majority of individuals will never develop cervical cancer following the procedure. It's more common for precancerous cells (CIN) to recur. This is not cancer, but it requires monitoring and sometimes further treatment. Recurrence of CIN is a natural reflection of persistent viral activity rather than a failure of the initial surgery. When abnormal cells reappear, they are almost always caught during routine screening at an early, highly manageable stage.
| Outcome After LEEP | Estimated Risk | What It Means |
|---|---|---|
| Recurrence of Precancerous Cells (CIN) | 5% - 10% | Abnormal cells have returned, requiring diligent follow-up and potentially more treatment. |
| Development of Invasive Cervical Cancer | 1% - 2% | The condition has progressed to cancer, which is a rare outcome after LEEP. |
These statistics underscore that while LEEP is highly effective, it is not a 100% cure. Your individual risk depends on several key factors identified during and after the procedure. It is also important to contextualize these numbers within the broader framework of modern cervical cancer screening. The 1-2% invasive cancer rate largely occurs in individuals who miss follow-up appointments or have untreated persistent high-risk HPV. With consistent co-testing and timely intervention, the risk of progression to invasive disease drops to nearly zero. Longitudinal studies following patients for 10 to 20 years post-LEEP demonstrate that those who adhere to surveillance protocols experience excellent oncologic outcomes and maintain normal life expectancy. Understanding absolute risk versus relative risk can also alleviate anxiety; while a 1-2% chance may sound concerning, it translates to a 98-99% probability of remaining cancer-free after the procedure.
It is worth noting that recurrence rates can vary depending on how "treatment failure" is defined in clinical literature. Some studies count any reappearance of low-grade changes (LSIL/CIN 1) as a recurrence, while others focus exclusively on high-grade dysplasia (HSIL/CIN 2+). The latter is the clinically relevant metric, as low-grade changes frequently regress spontaneously and rarely progress to cancer. Modern risk-based management guidelines utilize individualized risk thresholds to determine follow-up intensity. A patient with a 10-year post-treatment risk below 0.15% is generally managed with extended-interval screening, whereas higher risk thresholds prompt colposcopic evaluation. This personalized approach minimizes overtreatment while maintaining high sensitivity for detecting clinically significant lesions. Furthermore, the integration of biomarker testing, such as p16/Ki-67 dual staining, is increasingly being explored to triage patients with equivocal follow-up results. These advanced cytological markers can differentiate between true high-grade disease requiring intervention and transient cellular atypia that can be safely observed, thereby reducing unnecessary procedures and patient stress.
Key Factors That Influence Your Post-LEEP Risk
Understanding what influences the risk of recurrence can empower you to take a proactive role in your health. Research consistently points to two critical factors: surgical margins and HPV status. Additional clinical, behavioral, and biological variables also interact to shape your long-term risk profile.
Surgical Margin Status: Was It All Removed?
When the tissue is removed during a LEEP, a pathologist examines the edges, or "margins," under a microscope.
- Negative Margins: This is the ideal outcome. It means there are no abnormal cells at the edges of the removed tissue, suggesting the entire lesion was successfully excised.
- Positive Margins (or Involved Margins): This means abnormal cells were found at the edge of the tissue sample. It indicates that some precancerous cells may have been left behind on the cervix.
Positive margins are a significant predictor of recurrence. One large-scale study published in the Journal of Cancer Research and Clinical Oncology found that patients with positive margins had a 29.8% recurrence rate, compared to just 2.8% for those with negative margins. Pathologists often specify whether the involved margin is on the ectocervical (outer cervix) or endocervical (canal leading to the uterus) side. Endocervical involvement is generally considered more clinically significant because it can be more challenging to visualize and treat in future procedures. If margins are positive, your provider may recommend closer surveillance rather than immediate repeat surgery, as a significant portion of residual cells will regress spontaneously. In cases of high-grade disease (CIN 3) with positive margins, a repeat excision or hysterectomy may be discussed based on age, fertility goals, and patient preference.
Clinicians weigh margin status alongside the patient's HPV results and cytology at the 6-month post-treatment visit. Current evidence suggests that immediate re-excision for positive margins should be reserved for patients with high-grade disease on follow-up cytology or persistent HPV 16/18 positivity. For others, watchful waiting with co-testing at 6 and 12 months is often equally safe and spares patients from additional cervical trauma. The volume of tissue excised also matters; larger cone volumes correlate with wider negative margins but may increase obstetric risks. Surgeons therefore aim for an optimal balance, typically removing 10 to 15 mm of endocervical canal depth to achieve clear margins while minimizing cervical insufficiency. Advanced imaging techniques like 3D colposcopy and narrow-band imaging are being integrated into some specialty centers to improve pre-excision mapping, potentially reducing positive margin rates and optimizing therapeutic precision.
HPV Persistence: The Most Critical Factor
The LEEP procedure removes the cervical cells that have been damaged by the Human Papillomavirus (HPV), but it does not eliminate the virus from your body. Your immune system is responsible for clearing the underlying HPV infection. Persistent high-risk HPV (hr-HPV) infection is the single most important risk factor for the recurrence of CIN after LEEP. A 2024 meta-analysis provided compelling evidence:
- With negative margins and no detectable HPV after LEEP, the recurrence rate of CIN was a mere 0.5%.
- With negative margins but persistent HPV infection, the recurrence rate surged to 18%.
This highlights why follow-up HPV testing is just as important, if not more so, than the margin status. Modern clinical guidelines heavily weight HPV clearance as the primary biomarker for treatment success. Patients should understand that HPV clearance is a gradual process, typically taking 12 to 24 months post-procedure. The immune system relies on robust cell-mediated immunity to recognize and eliminate infected basal cells. Factors that support this clearance include adequate nutrition, stress management, sufficient sleep, and smoking cessation. Additionally, receiving the HPV vaccine after a LEEP is strongly supported by clinical evidence. Multiple studies demonstrate that vaccination in previously exposed individuals reduces the risk of CIN recurrence by approximately 40-50% by preventing new infections and potentially boosting cross-reactive immune responses against existing viral types.
HPV genotyping adds another layer of risk stratification. Infections with HPV 16 and 18 carry a significantly higher oncogenic potential compared to other high-risk types like HPV 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, or 68. Patients with persistent HPV 16/18 positivity require more vigilant surveillance due to the faster progression kinetics of these strains. Conversely, clearance of non-16/18 high-risk types often follows a more indolent course. Viral load quantification, though not yet standard in all guidelines, is increasingly utilized in research settings to gauge replication activity. High viral titers correlate with increased epithelial dysplasia and recurrence likelihood. Furthermore, viral integration into the host genome is a pivotal step toward malignant transformation. While excision removes integrated clones, ongoing surveillance detects reintegration events or new infections early. The advent of methylation markers, which detect epigenetic changes associated with viral oncogene expression, represents the next frontier in post-treatment monitoring, promising to identify high-risk patients before morphological changes become visible on cytology.
Other Contributing Risk Factors
Several other factors can play a role in your risk profile:
- High-Grade Lesion (CIN 3): Patients with more advanced precancerous changes (CIN 3) have a higher risk of recurrence than those with CIN 2.
- Age: Some studies suggest women over 40 may have a higher risk of recurrence, likely due to slower cellular turnover and diminished local immune response.
- Smoking: Smoking weakens the immune system's ability to fight HPV and is a known independent risk factor for both cervical cancer and post-LEEP recurrence. Nicotine and its metabolites concentrate in cervical mucus, directly damaging DNA and impairing Langerhans cell function.
- Weakened Immune System: Conditions like HIV or taking immunosuppressant drugs can make it harder for the body to clear HPV, increasing recurrence risk.
- Vaginal Microbiome Imbalance: Emerging research indicates that a healthy, lactobacillus-dominant vaginal microbiome protects against HPV persistence. Bacterial vaginosis and chronic inflammation can compromise the cervical epithelial barrier and reduce treatment efficacy.
- Co-infections: Sexually transmitted infections such as Chlamydia trachomatis or Herpes Simplex Virus can cause chronic mucosal inflammation, creating an environment that facilitates HPV integration and cellular dysplasia.
Beyond these well-established factors, nutritional status plays a surprisingly significant role in cervical healing and viral clearance. Deficiencies in folate, vitamin B12, vitamin C, vitamin D, and beta-carotene have been associated with impaired DNA repair mechanisms and prolonged HPV persistence. Folate is particularly crucial for maintaining genomic stability during rapid cellular regeneration post-excision. Supplementation or dietary optimization focusing on dark leafy greens, legumes, fortified grains, and antioxidant-rich fruits can create a biochemical environment hostile to viral replication. Additionally, metabolic health influences outcomes; insulin resistance and obesity are linked to chronic low-grade inflammation and altered estrogen metabolism, which can promote cervical epithelial proliferation and dysplasia. Weight management and glycemic control should therefore be considered integral components of post-LEEP care. Psychological stress also exerts measurable immunological effects. Chronic elevation of cortisol and catecholamines suppresses natural killer (NK) cell activity and T-helper 1 (Th1) cytokine production, both of which are essential for clearing intracellular pathogens like HPV. Incorporating stress-reduction modalities such as cognitive behavioral therapy, mindfulness meditation, or yoga has been shown in pilot studies to correlate with improved virological clearance rates and reduced anxiety surrounding follow-up visits.
Understanding risk factors is key to managing health after a LEEP. Source: Thomson Medical.
Life After LEEP: Recovery, Follow-Up, and Long-Term Health
Your care doesn't end when the procedure is over. The post-LEEP period is crucial for healing and long-term prevention. Managing expectations, adhering to post-operative instructions, and maintaining open communication with your gynecologist will optimize your recovery and surveillance outcomes. This transitional phase bridges acute tissue repair with the establishment of lifelong preventive health habits.
Navigating the Recovery Period
Recovery typically takes about four weeks. During this time, you can expect:
- Mild cramping, similar to a menstrual period.
- Watery vaginal discharge, which may be pinkish or have a slight odor.
- A dark, coffee-ground-like discharge, which is from the medicated paste used to control bleeding.
To ensure proper healing, your doctor will advise you to avoid the following for at least four weeks:
- Vaginal intercourse
- Using tampons or menstrual cups (use pads instead)
- Douching
- Heavy lifting or strenuous exercise
- Bathing or swimming (showers are fine)
These precautions are designed to prevent introducing bacteria into the healing cervical wound and to avoid disrupting the eschar (scab) that forms over the excision site. You can typically return to light daily activities and desk work within 24 to 48 hours. Gradually reintroduce moderate exercise after two weeks, but avoid high-impact activities or heavy lifting (over 20 pounds) until the four-week mark. Over-the-counter analgesics like ibuprofen are usually sufficient for cramping. Applying a heating pad to the lower abdomen can provide additional comfort. Contact your doctor if you experience severe abdominal pain, heavy bleeding (soaking more than one pad an hour), fever, or foul-smelling discharge, as these could be signs of an infection or complication. While rare, delayed hemorrhage can occur 10-14 days post-procedure as the cauterized tissue sloughs off. In such cases, medical evaluation is necessary to determine if additional hemostatic measures are required. Another potential, though uncommon, long-term effect is cervical stenosis (narrowing of the cervical canal), which may cause lighter periods, difficulty with future Pap smears, or mild dysmenorrhea. If you notice significant changes in your menstrual flow or difficulty conceiving later, discuss this with your provider, as minor office-based dilation can often resolve the issue.
Hydration and nutritional support accelerate tissue regeneration. Drinking adequate water helps thin vaginal secretions, making clearance more comfortable and reducing the risk of secondary bacterial overgrowth. Consuming lean proteins, zinc-rich foods, and vitamin C supports collagen synthesis and angiogenesis at the excision site. It is also advisable to avoid non-steroidal anti-inflammatory drugs (NSAIDs) like naproxen immediately before the procedure, as they can affect platelet function, but they remain excellent choices for post-procedure pain control. Wearing breathable, cotton underwear and using unscented panty liners can maintain local dryness and prevent irritation from discharge. Many patients experience emotional fluctuations during recovery due to hormonal shifts, surgical stress, and health-related anxiety. Keeping a symptom journal, joining a peer support network, or scheduling a follow-up telehealth check-in at the two-week mark can provide reassurance and catch any complications early. Remember that each person's healing timeline is unique; comparing your recovery to others online may unnecessarily heighten stress.
The Importance of Long-Term Surveillance
A LEEP is not a "one-and-done" treatment. Consistent, long-term follow-up is non-negotiable and the most important step you can take to protect your health. Current guidelines, such as those from the American Society for Colposcopy and Cervical Pathology (ASCCP), recommend co-testing (a Pap test and an HPV test) 6 months after the LEEP procedure.
- If these tests are negative, you will likely continue with regular screenings.
- If either test is positive, your doctor may recommend a colposcopy to investigate further.
This surveillance schedule is crucial for catching any persistent or recurrent disease early, when it is most treatable. Following the initial 6-month test, patients typically transition to co-testing at 12 months and then again at 18-24 months. Once three consecutive annual tests are negative, you can generally return to a 3-year co-testing or 5-year HPV-only screening interval, depending on age and local guidelines. Importantly, individuals with a history of high-grade CIN are usually advised to continue cervical cancer screening for at least 20 to 25 years post-treatment, even past the age of 65, because the long-term risk remains slightly elevated compared to the general population. Some healthcare systems are now integrating patient-initiated self-sampling for HPV, which can improve adherence and reduce barriers to routine monitoring.
The shift toward risk-based management has revolutionized post-treatment surveillance. Instead of rigid calendar-based schedules, clinicians now utilize validated risk calculators that incorporate histology, margin status, HPV genotype, and cytology to generate personalized screening intervals. For low-risk patients, extending intervals to every three years reduces overtreatment and anxiety without compromising safety. High-risk patients, particularly those with positive margins or persistent HPV 16, may undergo more frequent colposcopy or adjunctive testing. Primary HPV testing is increasingly favored over Pap-smear-only surveillance due to its higher negative predictive value. A negative HPV test post-treatment is one of the strongest reassurances that recurrence risk has plummeted to baseline levels. Patients should maintain a personal health record or use patient portal apps to track their screening dates, results, and upcoming appointments. Setting automated reminders and advocating for same-day results can streamline the process and ensure timely clinical decisions. Accessibility remains a barrier for some, so discussing transportation assistance, workplace accommodation for medical leave, or telehealth result consultations with your care team is highly encouraged.
What Happens if Abnormal Cells Return?
If follow-up testing reveals that high-grade dysplasia has persisted or recurred, your doctor will discuss the next steps. This is known as an escalation of care. The options depend on the severity of the cells, your age, and your desire for future pregnancies.
- Repeat LEEP: For many, especially those who wish to preserve fertility, a second LEEP procedure can be performed to remove the remaining abnormal tissue. Providers take care to limit the amount of tissue removed to preserve cervical competency.
- Alternative Ablative or Excisional Therapies: Depending on the anatomy of the recurrence, laser conization or cryotherapy may be considered.
- Hysterectomy: For women who have completed childbearing or when a repeat excision is not a suitable option, a hysterectomy (surgical removal of the uterus and cervix) is a definitive treatment that eliminates the risk of cervical cancer.
- Fertility and Pregnancy Considerations: It is natural to worry about how a LEEP affects future pregnancies. Research indicates that while LEEP is associated with a modest increase in the risk of preterm birth and premature rupture of membranes, the absolute risk remains low. The risk correlates with the volume of tissue removed. If you become pregnant after a LEEP, your obstetrician will likely monitor your cervical length via transvaginal ultrasound in the second trimester. If shortening is detected, interventions like cervical cerclage or vaginal progesterone can be highly effective in prolonging gestation.
When recurrence is identified, a multidisciplinary approach is often beneficial. Gynecologic oncologists, maternal-fetal medicine specialists, and reproductive endocrinologists may collaborate to balance oncologic safety with fertility preservation. For patients pursuing a repeat excision, cold knife conization (CKC) is sometimes preferred if the lesion extends high into the endocervical canal or if precise margin assessment is critical. CKC utilizes a scalpel rather than electrosurgery, eliminating thermal artifact but requiring anesthesia in an operating room setting. In contrast, ablative techniques like laser therapy destroy dysplastic tissue without obtaining a specimen, making them suitable only when invasive cancer has been definitively ruled out via biopsy. Psychological support should also be considered when managing recurrence or navigating long-term surveillance. The anxiety associated with repeated testing is valid and common. Many patients find reassurance in connecting with support groups, practicing mindfulness techniques, or speaking with a counselor who specializes in women's health. Remember that recurrence is a manageable clinical pathway, not a personal failure. Modern oncology protocols ensure that even persistent disease is intercepted well before it reaches an invasive stage.
Conclusion: A Proactive Approach to Your Cervical Health
Receiving a diagnosis of cervical dysplasia and undergoing a LEEP can be a stressful experience, but it's important to view it as a proactive and highly effective step in preventing cancer. The chance of developing cervical cancer after a LEEP is very low. Your long-term health depends on a partnership between you and your healthcare provider. By understanding the key risk factors like HPV persistence and adhering strictly to your recommended follow-up schedule, you can significantly minimize your risk and ensure any potential issues are addressed immediately. Embrace lifestyle modifications that support immune function, stay up to date with the HPV vaccine if recommended, and maintain open communication about your reproductive goals and concerns. Modern gynecology has transformed cervical dysplasia from a life-threatening uncertainty into a highly manageable, routinely screened condition. You are not defined by a precancer diagnosis; rather, you are empowered by the knowledge that you've taken decisive action to protect your future.
As cervical cancer screening and treatment continue to evolve, patient education remains the most potent tool in your arsenal. Advances in molecular diagnostics, minimally invasive surgical techniques, and personalized risk algorithms mean that post-LEEP care is more precise and less disruptive than ever before. By staying informed, asking questions, and advocating for your health, you position yourself to thrive long after the procedure. Trust the process, honor your body's healing timeline, and recognize that each follow-up visit is a milestone of resilience and prevention. With consistent care and proactive habits, the vast majority of individuals successfully transition back to routine wellness monitoring, fully confident in their long-term health outlook.
Frequently Asked Questions
Can I still have a healthy pregnancy after a LEEP procedure?
Yes, the vast majority of women who undergo a LEEP go on to have healthy, full-term pregnancies. The procedure is designed to preserve reproductive function by targeting only the abnormal tissue while leaving healthy cervical structure intact. However, removing cervical tissue can slightly weaken the cervix, which marginally increases the risk of preterm labor or low birth weight. If you plan to conceive after your procedure, it is generally recommended to wait at least 3 to 6 months to allow the cervix to fully heal. During pregnancy, your healthcare provider will monitor your cervical length via transvaginal ultrasound in the second trimester. If shortening is detected, interventions like progesterone therapy or cervical cerclage can be highly effective in preventing premature delivery. Open dialogue with your obstetrician about your surgical history ensures tailored prenatal care.
How long do I need to continue more frequent Pap smears after treatment?
Following a LEEP for high-grade dysplasia, you will typically transition into an intensified surveillance schedule. This usually involves co-testing (Pap and HPV) at 6 months, 12 months, and 24 months post-procedure. If all tests remain negative during this period, you can often return to standard screening intervals (every 3 to 5 years, depending on age and guidelines). However, because your personal history places you at a slightly higher baseline risk than the general population, most clinical guidelines recommend continuing routine cervical cancer screening for at least 25 years after treatment, even if you reach the age of 65. Your provider will tailor the exact timeline based on your pathology results and HPV clearance status. Adhering to this extended surveillance window dramatically reduces the likelihood of missing late recurrences.
Does getting the HPV vaccine after LEEP provide any benefit?
Absolutely. Clinical evidence strongly supports HPV vaccination even after you have already been exposed to the virus or undergone a LEEP. While the vaccine is not therapeutic for your existing infection, it significantly reduces the risk of reinfection with other high-risk HPV strains and lowers the likelihood of cervical dysplasia recurrence by up to 50%. The immune response generated by the vaccine may also cross-react to help clear the currently active infection through enhanced antibody production and T-cell activation. Patients up to age 45 are generally eligible for vaccination, and many gynecologists now routinely recommend it as part of comprehensive post-LEEP care to maximize long-term protection. Shared decision-making with your clinician will determine the optimal timing and dosing schedule.
What lifestyle changes can I make to help my body clear HPV faster?
HPV clearance is largely driven by your cell-mediated immune system, so supporting overall health is crucial. Quitting smoking is the single most impactful lifestyle change, as tobacco byproducts directly suppress local cervical immunity and impair viral clearance. Prioritize a balanced diet rich in folate, vitamin B12, antioxidants, and cruciferous vegetables like broccoli and kale, which have been associated with healthier cervical epithelium and DNA repair mechanisms. Regular moderate exercise, stress reduction techniques (such as meditation or yoga), and aiming for 7-9 hours of quality sleep each night will bolster immune function. Additionally, maintaining a healthy vaginal microbiome by avoiding douching, using gentle, pH-balanced cleansers externally only, and wearing breathable cotton underwear can reduce chronic inflammation and support local defense mechanisms against viral persistence.
Is it safe to resume sexual activity immediately after I feel fine?
You should wait until your healthcare provider explicitly clears you, which is typically at the 4-week follow-up appointment. Even if external symptoms like discharge or cramping have resolved, the internal cervical tissue is still undergoing epithelial regeneration and scab shedding during this period. Resuming intercourse, using tampons, or placing anything in the vagina too early can introduce bacteria, disrupt the healing wound, trigger delayed bleeding, or increase the risk of pelvic inflammatory disease. When you do resume sexual activity, use barrier protection like condoms initially to reduce the risk of re-exposing the healing cervix to other sexually transmitted infections, which could interfere with HPV clearance. Communication with your partner about temporary modifications and comfort measures can ease the transition back to intimacy.
What should I do if my insurance won't cover my follow-up colposcopy or HPV testing?
Consistent follow-up is critical, and there are several pathways to access affordable care. Federally Qualified Health Centers (FQHCs) and county public health clinics offer sliding-scale fees for cervical cancer screening and surveillance based on income. The National Breast and Cervical Cancer Early Detection Program (NBCCEDP) provides free or low-cost Pap smears, HPV testing, and diagnostic follow-up for qualifying uninsured or underinsured individuals in all 50 states. Additionally, many laboratory companies offer patient assistance programs for self-pay testing, and organizations like the American Cancer Society or local women's health nonprofits can help navigate financial assistance applications. Do not skip appointments due to cost; speak with your provider's billing department or a medical social worker to explore these resources. Preventive care investments now avert significantly higher medical expenses down the line.
References
- Medical News Today. (2023). Odds of cervical cancer after LEEP: Effectiveness and more. Retrieved from https://www.medicalnewstoday.com/articles/chances-of-cervical-cancer-after-leep
- Healthline. (2024). Chances of Cervical Cancer After a LEEP Procedure. Retrieved from https://www.healthline.com/health/cervical-cancer/chances-of-cervical-cancer-after-leep
- Cleveland Clinic. (2022). Loop Electrosurgical Excision Procedure (LEEP). Retrieved from https://my.clevelandclinic.org/health/treatments/4711-loop-electrosurgical-excision-procedure-leep
- Ding, T., et al. (2022). Risk factors analysis of recurrent disease after treatment with a loop electrosurgical excision procedure in patients with cervical intraepithelial neoplasia 2–3: a retrospective study of 4369 patients. Journal of Cancer Research and Clinical Oncology. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC10087663/
- Bogani, G., et al. (2020). Recurrence rate after loop electrosurgical excision procedure (LEEP) and laser Conization: A 5-year follow-up study. Gynecologic Oncology. Retrieved from https://www.sciencedirect.com/science/article/abs/pii/S0090825820338269
- Johns Hopkins Medicine. Fertility and Pregnancy After a LEEP. Retrieved from https://www.hopkinsmedicine.org/health/wellness-and-prevention/fertility-and-pregnancy-after-a-leep

About the author
Sofia Rossi, MD, is a board-certified obstetrician-gynecologist with over 15 years of experience in high-risk pregnancies and reproductive health. She is a clinical professor at a top New York medical school and an attending physician at a university hospital.