Understanding the Cost of BRCA Gene Testing

Key points
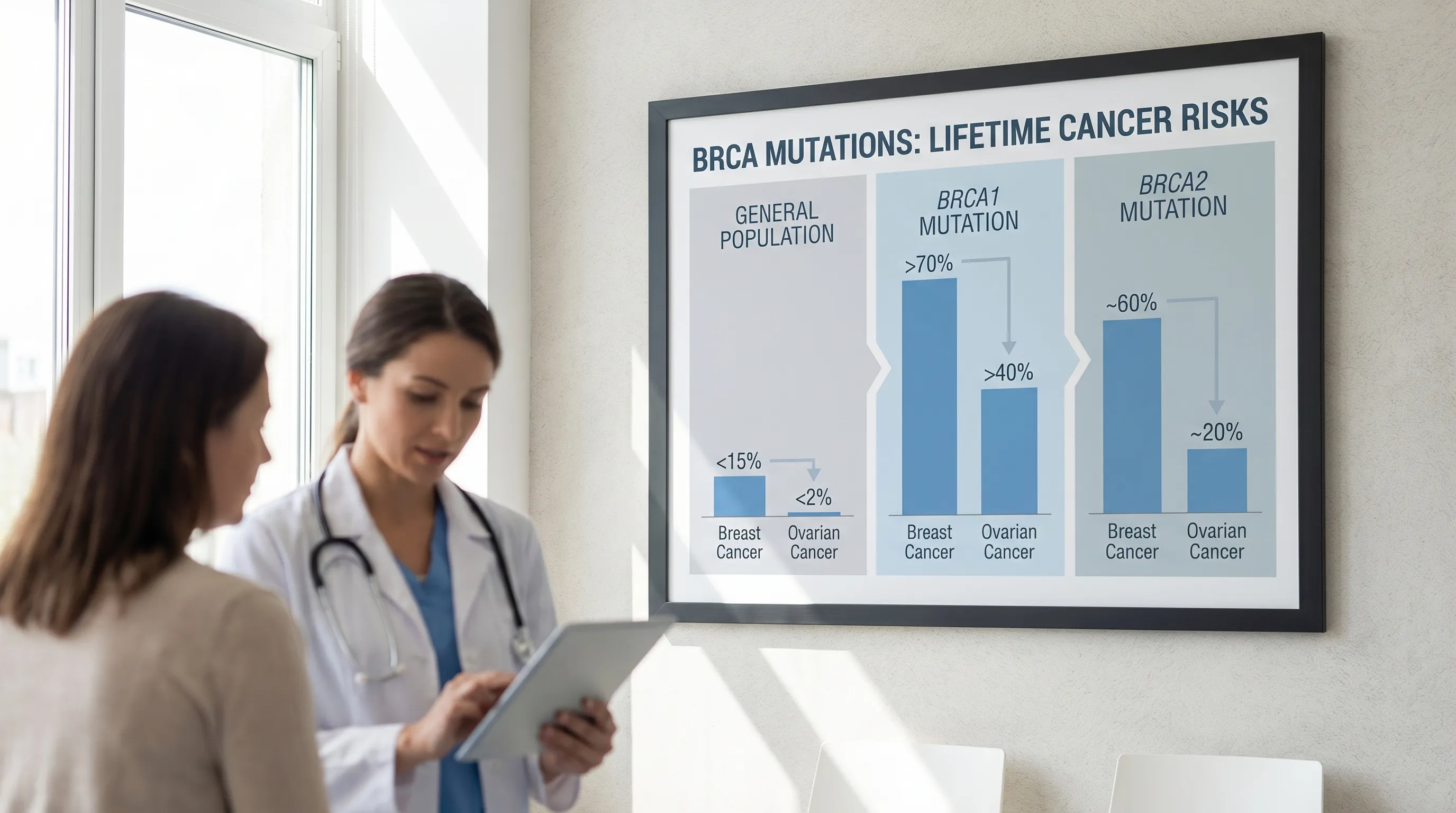
- BRCA1 Mutation: Significantly increases a woman's lifetime risk of breast cancer to 65-75% and ovarian cancer to 40-45%. Men also have an increased risk of breast and pancreatic cancer. BRCA1-related breast cancers are more frequently diagnosed at younger ages and are disproportionately likely to be triple-negative breast cancer (TNBC), a subtype that lacks estrogen, progesterone, and HER2 receptors, making it more aggressive and less responsive to traditional hormonal therapies.
- BRCA2 Mutation: Increases a woman's lifetime risk of breast cancer to 45-70% and ovarian cancer to 10-20%. Men with BRCA2 mutations have a higher risk of prostate, breast, and pancreatic cancer. BRCA2-associated breast cancers more often present with hormone receptor-positive features, but they still carry a substantially higher baseline risk than sporadic cases. BRCA2 mutations are also strongly linked to early-onset aggressive prostate cancer and melanoma.
BRCA gene testing can be a powerful tool for understanding your risk for certain hereditary cancers. Prompted by high-profile stories like Angelina Jolie's decision to undergo preventive surgery, awareness has grown around the BRCA1 and BRCA2 genes. Harmful mutations in these genes significantly increase the lifetime risk of developing breast, ovarian, prostate, and pancreatic cancers. Hereditary breast and ovarian cancer (HBOC) syndrome, which is primarily driven by these mutations, accounts for approximately 5% to 10% of all breast cancer cases and up to 15% of ovarian cancer cases. In the general population, about 1 in 400 to 1 in 800 individuals carry a pathogenic BRCA variant, but this prevalence rises dramatically to roughly 1 in 40 among individuals of Ashkenazi Jewish descent due to founder mutations. Recognizing these statistical realities underscores why genetic literacy and proactive screening have become cornerstones of modern preventive oncology.
Understanding the process, implications, and especially the cost of testing is crucial for making an informed decision. This guide breaks down everything you need to know about the cost of BRCA gene testing, from insurance coverage to affordable self-pay options, while also providing clinical context to help you navigate the healthcare landscape with confidence.

What Are BRCA Genes and Why is Testing Important?
BRCA1 and BRCA2 are tumor suppressor genes that produce proteins to repair damaged DNA. When a harmful mutation exists in one of these genes, this repair function is impaired, allowing DNA damage to accumulate, which can lead to cancer. Specifically, BRCA1 and BRCA2 play an essential role in the homologous recombination pathway, a high-fidelity mechanism that fixes double-strand DNA breaks. Without fully functional BRCA proteins, cells rely on error-prone backup repair methods, dramatically increasing the likelihood of chromosomal instability and malignant transformation. This biological vulnerability is why carriers face significantly elevated cancer risks across multiple organ systems.
- BRCA1 Mutation: Significantly increases a woman's lifetime risk of breast cancer to 65-75% and ovarian cancer to 40-45%. Men also have an increased risk of breast and pancreatic cancer. BRCA1-related breast cancers are more frequently diagnosed at younger ages and are disproportionately likely to be triple-negative breast cancer (TNBC), a subtype that lacks estrogen, progesterone, and HER2 receptors, making it more aggressive and less responsive to traditional hormonal therapies.
- BRCA2 Mutation: Increases a woman's lifetime risk of breast cancer to 45-70% and ovarian cancer to 10-20%. Men with BRCA2 mutations have a higher risk of prostate, breast, and pancreatic cancer. BRCA2-associated breast cancers more often present with hormone receptor-positive features, but they still carry a substantially higher baseline risk than sporadic cases. BRCA2 mutations are also strongly linked to early-onset aggressive prostate cancer and melanoma.
Knowing you carry a mutation allows you and your healthcare team to create a personalized plan for managing your cancer risk. This may include enhanced screening, risk-reducing medications, or preventive surgeries, all of which can dramatically improve long-term health outcomes. Importantly, genetic risk does not equate to a cancer diagnosis; penetrance is influenced by environmental factors, lifestyle choices, and other genetic modifiers. Regular mammography and breast MRI, transvaginal ultrasound, CA-125 blood testing, and chemoprevention strategies like tamoxifen or raloxifene are all evidence-based interventions tailored to mutation carriers.
“Having a BRCA gene mutation significantly increases a person’s risk for breast and ovarian cancers, but knowledge is power – with this information, individuals can take proactive steps for early detection or risk reduction.”
– Susan Domchek, MD, Executive Director of the Basser Center for BRCA at Penn Medicine
Who Should Consider BRCA Gene Testing?
Genetic testing for BRCA mutations is not recommended for everyone. It is typically advised for individuals with personal or family health histories that suggest a higher likelihood of carrying a mutation. According to guidelines from organizations like the BRCA Genetic Testing and Guidelines, you should consider testing if you have:
- A personal history of breast cancer (especially if diagnosed before age 50, triple-negative, or in both breasts), ovarian cancer, pancreatic cancer, or metastatic prostate cancer.
- A family history of a known BRCA1 or BRCA2 mutation.
- Multiple relatives on the same side of the family with breast, ovarian, pancreatic, or prostate cancer.
- A male relative with breast cancer.
- Ashkenazi Jewish ancestry, as specific BRCA mutations are more common in this population.
- Additional red flags include bilateral breast cancer, breast and ovarian cancer in the same individual, ovarian or peritoneal cancer at any age, or a combination of pancreatic and prostate cancers across first- or second-degree relatives.
Genetic testing is generally not recommended for minors unless there is a childhood-onset condition linked to the mutation, which does not apply to BRCA. Testing is reserved for adults who can provide informed consent and are emotionally prepared to act on the results.
Consulting with a doctor or genetic counselor is the best first step to assess your personal risk and determine if testing is right for you. A certified genetic counselor will construct a detailed pedigree spanning at least three generations, calculate your pre-test probability of harboring a pathogenic variant, and discuss the psychological, medical, and financial implications before ordering any lab work.
The BRCA Gene Testing Process
The journey from consideration to results typically involves several key steps:
- Genetic Counseling: A genetic counselor will review your personal and family medical history, discuss the pros and cons of testing, and explain what the results might mean for you and your family. This session typically lasts 30 to 60 minutes and is considered a critical component of the testing protocol. Many insurers require documented pre-test counseling before approving coverage.
- Sample Collection: Testing is done using a simple blood draw or a saliva sample. Blood is traditionally processed in CLIA-certified laboratories and provides high-quality genomic DNA. Saliva kits, increasingly popular for convenience, contain stabilizing buffers that preserve DNA during shipping. Both methods yield clinically equivalent results for BRCA analysis when processed by accredited labs.
- Laboratory Analysis: Your DNA is analyzed in a specialized lab to look for mutations in the BRCA1 and BRCA2 genes. Many tests today are part of multi-gene panels, which check other cancer-related genes simultaneously. Modern next-generation sequencing (NGS) technology allows labs to sequence millions of DNA fragments in parallel, identifying single nucleotide variants (SNVs), small insertions, and deletions. Labs often combine sequencing with deletion/duplication analysis (MLPA) to catch larger structural rearrangements.
- Receiving Results: Results typically take two to four weeks. They will be classified as positive, negative, or a "variant of uncertain significance" (VUS). A positive result confirms a pathogenic or likely pathogenic variant. A true negative means no clinically significant mutation was found in the genes tested. A VUS indicates a genetic change whose impact on cancer risk is currently unknown; it is not used to guide clinical management and is periodically reclassified as research advances.
- Post-Test Counseling: A healthcare professional will help you understand your results and discuss the next steps for managing your health, whether that involves increased screening, preventive measures, or informing family members. Post-test counseling ensures patients understand the difference between carrier status and disease, reviews cascade testing for relatives, and establishes a long-term surveillance or risk-reduction plan.
Understanding the Cost of BRCA Gene Testing
The cost of BRCA testing has decreased significantly since a 2013 Supreme Court ruling ended gene patenting, allowing for more competition among labs. However, the price can still vary widely depending on a complex matrix of clinical, administrative, and geographic factors. Before the 2013 Association for Molecular Pathology v. Myriad Genetics decision, a single BRCA test could cost upwards of $3,000 to $4,000 with little to no pricing transparency. Today, the market is more competitive, but navigating the financial landscape still requires diligence.
Factors Influencing the Cost
- Type of Test: A targeted test for a known family mutation is less expensive than comprehensive sequencing of the entire BRCA1 and BRCA2 genes. If a relative already has an identified variant, "single-site testing" only looks for that specific mutation, drastically reducing sequencing time and cost.
- Multi-Gene Panels: Many labs now offer panels that test for dozens of cancer-risk genes. Surprisingly, these are often similarly priced to BRCA-only tests. Panels often include genes like CHEK2, PALB2, ATM, and TP53, which are associated with overlapping hereditary cancer syndromes. The added value of a panel must be weighed against the increased likelihood of encountering VUS results.
- Insurance Coverage: Whether your insurance covers the test is the single biggest factor in your out-of-pocket cost. Coverage hinges on medical necessity, which is determined by your family/personal history, your insurer’s specific clinical policy, and whether proper coding and prior authorization were submitted.
- Lab Choice: Prices differ between laboratories like Invitae, Myriad Genetics, LabCorp, Ambry Genetics, and others. Labs negotiate different contracted rates with insurance networks, and self-pay pricing varies based on their operational models, marketing strategies, and volume discounts.
- Additional Services: The cost may or may not include genetic counseling sessions, which can be billed separately. Counseling can cost between $150 and $500 per session depending on the provider's credentials, geographic location, and insurance contract status. Some direct-to-consumer or lab-sponsored programs bundle counseling into a flat cash price.
Typical Cost Ranges (With and Without Insurance)
- With Insurance Coverage: If you meet your insurer's criteria for medical necessity, your out-of-pocket cost may be $0 or limited to a small copay or deductible. The Affordable Care Act (ACA) mandates that preventive services, including BRCA testing for high-risk women, be covered with no cost-sharing. This applies to USPSTF Grade A and B recommendations, which include genetic risk assessment and counseling for women with a family history suggestive of BRCA mutations. However, the testing itself is often covered under diagnostic benefits, which may apply toward your deductible.
- Without Insurance (Self-Pay): If you pay out-of-pocket, the cost can range from $250 to over $3,000. Many leading labs have established a self-pay price of around $250-$450 for a comprehensive hereditary cancer panel. This aggressive pricing shift reflects industry competition and a desire to remove financial barriers that delay critical risk stratification. Always request a written good-faith estimate before testing, as required by the No Surprises Act.
- Direct-to-Consumer (DTC) Tests: Companies like 23andMe offer limited BRCA testing as part of their health reports for around $200. Crucially, these tests only screen for a few specific mutations (typically the three Ashkenazi Jewish founder mutations) and are not comprehensive. They are not a substitute for clinical-grade testing if you have significant risk factors, as a negative DTC result does not rule out hundreds of other pathogenic variants. Clinical confirmation of any positive DTC result in a CLIA-certified lab is standard medical practice before making any treatment decisions.
Does Insurance Cover BRCA Gene Testing?
For many people, insurance is the key to affordable testing. Navigating the pre-authorization process successfully requires understanding how insurers evaluate genetic claims.
- Private Insurance: Most plans cover BRCA testing if you meet specific criteria based on your personal or family cancer history, as defined by the NCCN or USPSTF. Pre-authorization is often required. Insurers typically review documentation from your ordering physician, including your family pedigree, pathology reports, and a letter of medical necessity. Using an in-network laboratory is critical; out-of-network testing can trigger balance billing and full financial responsibility.
- Medicare: Covers genetic testing for individuals who have already been diagnosed with a cancer linked to BRCA and meet specific criteria. Coverage for unaffected individuals based solely on family history can be limited, though Medicare has expanded coverage in recent years to include testing for asymptomatic beneficiaries when it will directly impact management. Part B typically covers the lab test and counseling with 20% coinsurance after the deductible is met.
- Medicaid: Coverage varies by state, but many programs cover testing for individuals who meet high-risk criteria. Some states require step-therapy approaches, such as testing an affected relative first before covering an unaffected family member. Always verify benefits through your state's Medicaid managed care portal or by calling the member services number on your card.
International Cost and Coverage
Coverage and cost differ significantly around the world. Healthcare system structures, centralized genetic services, and national screening programs heavily influence patient out-of-pocket expenses.
| Country/Region | Approximate Out-of-Pocket Cost (USD) | Coverage Details |
|---|---|---|
| United States | $0 (with insurance) to $250-$3,000+ | Most insurance plans cover testing for high-risk individuals. ACA preventive mandate applies to genetic counseling and BRCA assessment for eligible women. |
| Canada | $750 - $1,500 | Covered by provincial healthcare if risk criteria are met. Wait times for publicly funded genetic counseling can range from months to over a year, prompting some to seek private testing. |
| United Kingdom | $1,300 - $2,000 | Covered by the NHS for eligible individuals through regional medical genetics centers; private testing available. NHS England follows strict eligibility criteria aligned with NICE guidelines. |
| Australia | $400 - $800 | Covered by Medicare for high-risk individuals when ordered through a recognized genetics clinic. Medicare Benefits Schedule (MBS) item numbers exist for specific hereditary cancer testing pathways. |
Costs are estimates and may vary. Always verify with local providers. Exchange rates and public healthcare funding adjustments can shift these figures annually.
Financial Assistance and Low-Cost Options
If cost is a barrier, several options are available:
- Lab Financial Assistance: Many testing labs have patient assistance programs or payment plans. Some cap the out-of-pocket cost at a few hundred dollars. Companies frequently offer sliding-scale fees based on household income, household size, and documented insurance denial letters.
- Non-Profit Organizations: Groups like FORCE (Facing Our Risk of Cancer Empowered) and Bright Pink provide resources and may be aware of assistance programs. FORCE also maintains a financial aid directory specifically for high-risk hereditary cancer populations, including grants for genetic counseling and testing.
- Research Studies: You may be eligible for free testing by participating in a clinical trial. Check ClinicalTrials.gov for relevant studies. Academic medical centers frequently recruit unaffected carriers or newly diagnosed patients for hereditary cancer registries, which often include complimentary comprehensive genomic testing and longitudinal follow-up.
- Health Savings Accounts (HSAs) & Flexible Spending Accounts (FSAs): BRCA testing qualifies as an eligible medical expense under IRS Publication 502. Using pre-tax dollars from an HSA or FSA can effectively reduce your out-of-pocket cost by 20-30%, depending on your marginal tax bracket. Ensure you keep the itemized receipt and a letter of medical necessity for tax documentation.
Navigating Emotional and Legal Considerations
The "cost" of testing extends beyond the financial price tag. The psychological and administrative dimensions of genetic testing are just as critical to long-term well-being and should be factored into your decision-making process.
- Emotional Impact: Waiting for results can be stressful, and a positive result can bring a mix of relief (from uncertainty) and anxiety. Support groups and counseling are valuable resources. Some individuals experience "scanxiety" leading up to heightened surveillance appointments, while others feel empowered by actionable data. It is normal to grieve the loss of perceived health security, but genetic literacy programs and peer mentorship have been shown to significantly reduce psychological distress post-testing.
- Genetic Discrimination: In the U.S., the Genetic Information Nondiscrimination Act (GINA) prevents health insurers and most employers from discriminating based on your genetic information. However, GINA does not apply to life, disability, or long-term care insurance. It also does not cover members of the U.S. military, individuals receiving care through the Indian Health Service, or employers with fewer than 15 employees. Many experts strongly recommend securing life and long-term care insurance policies before undergoing genetic testing to avoid potential premium increases or coverage exclusions later.
- Family Implications: Your result has implications for your relatives. Genetic counselors can provide guidance on how to share this sensitive information with family members who may also be at risk. Cascade testing—systematically offering genetic evaluation to biological relatives once a mutation is identified in a family—is a proven public health strategy. While some patients feel hesitant to initiate conversations about hereditary cancer, providing relatives with a provider-generated summary letter can ease communication and facilitate independent clinical consultations.
- Privacy and Data Security: Genetic data is inherently sensitive and uniquely identifiable. Reputable CLIA-certified labs adhere to HIPAA regulations, but it is important to read the privacy policies of any testing company. Understand who owns the de-identified genomic data, whether it can be sold to pharmaceutical or biotechnology firms for research, and how to request permanent data deletion if permitted. Always opt for clinically accredited laboratories rather than unregulated wellness platforms when medical decision-making is the goal.
Additional Resources
For more information, consult these trusted sources:
- National Cancer Institute: BRCA Gene Mutations: Cancer Risk and Genetic Testing
- American Cancer Society: Genetic Testing for Cancer Risk
- Mayo Clinic: BRCA gene test
- Find a Genetic Counselor: National Society of Genetic Counselors
- Genetic and Rare Diseases (GARD) Information Center: Hereditary Cancer Syndromes
- FORCE (Facing Our Risk of Cancer Empowered): Patient Advocacy and Education
- Centers for Medicare & Medicaid Services (CMS): Coverage for Genetic Testing Services
Utilizing these resources can help demystify the genetic testing landscape, clarify insurance navigation strategies, and connect you with patient advocacy networks. Many organizations offer downloadable toolkits for family history collection, insurance appeal letter templates, and pre-consultation questionnaires to maximize your clinical encounters.
Frequently Asked Questions
Can my health insurance company drop me or raise my premiums after a positive BRCA test?
No, not your health insurance. Under the Genetic Information Nondiscrimination Act (GINA), group and individual health insurers are legally prohibited from denying coverage, charging higher premiums, or imposing pre-existing condition exclusions based on genetic test results. However, GINA's protections do not extend to life insurance, disability insurance, or long-term care insurance. These providers may legally request your medical and genetic records during the underwriting process and adjust premiums or deny coverage based on elevated hereditary risk.
What happens if my test comes back as a Variant of Uncertain Significance (VUS)?
A VUS means a genetic change was found, but current scientific evidence is insufficient to classify it as either disease-causing or benign. Clinically, a VUS is treated as a negative result for management purposes, meaning you should continue standard population-based cancer screening. Labs periodically re-evaluate VUS classifications as global research and variant databases expand. If reclassification occurs, the lab typically contacts your ordering provider, who will then reach out to you to update your care plan.
Should I pay out-of-pocket or try to use my insurance if I meet the criteria?
Both approaches have pros and cons. Using insurance often results in $0 out-of-pocket cost but requires navigating prior authorization, using an in-network lab, and accepting the permanent inclusion of a genetic test diagnosis code in your medical record. Paying out-of-pocket offers privacy and avoids insurance bureaucracy, and many reputable labs now offer flat cash rates between $250 and $450 for comprehensive panels. The choice depends on your privacy preferences, financial situation, and comfort level with insurance documentation. Always verify network status and obtain a written estimate before proceeding.
Is it too late to get tested if I've already been diagnosed with cancer?
Absolutely not. In fact, testing after a cancer diagnosis can be highly clinically valuable. BRCA mutation status directly influences treatment options, particularly the use of PARP inhibitors (like olaparib or talazoparib) and platinum-based chemotherapy, which are especially effective in BRCA-associated cancers due to synthetic lethality in DNA repair pathways. Additionally, a positive result informs surgical decisions (such as opting for a double mastectomy over a lumpectomy) and guides screening and prevention strategies for surviving family members through cascade testing.
How accurate are direct-to-consumer BRCA tests, and can I use them instead of clinical testing?
DTC tests are highly accurate for the specific variants they screen for, but their clinical utility is extremely limited. Most DTC platforms only check for three founder mutations common in Ashkenazi Jewish populations. They do not sequence the entire BRCA1 or BRCA2 genes, meaning a negative DTC result provides false reassurance if you carry a non-founder mutation. If a DTC test returns a positive result, clinical confirmation via a CLIA-certified laboratory is mandatory before making any medical or surgical decisions. For individuals with significant family history or non-Ashkenazi ancestry, clinical-grade testing ordered by a healthcare professional remains the gold standard.
Conclusion
BRCA gene testing represents a critical intersection of personalized medicine, preventive care, and financial navigation. While the upfront cost of testing can seem daunting, the landscape has evolved dramatically to prioritize patient access, with robust insurance mandates, competitive self-pay pricing, and comprehensive financial assistance programs ensuring that cost is rarely a permanent barrier to knowledge. Understanding the clinical implications of BRCA1 and BRCA2 mutations, from hereditary breast and ovarian cancer syndrome risks to targeted therapeutic options like PARP inhibitors, empowers patients and providers to make proactive, life-altering health decisions.
Successfully navigating the testing process requires more than just financial planning; it demands thorough pre- and post-test genetic counseling, careful consideration of privacy and legal protections, and open communication within families. Whether you choose to pursue clinical-grade testing through your insurance, utilize an accredited laboratory's self-pay pricing, or explore research-based avenues, the ultimate goal remains the same: early risk stratification, tailored surveillance, and improved long-term outcomes. By leveraging the resources, guidelines, and professional support available today, you can confidently take control of your genetic health journey and make informed choices that align with your medical needs, financial reality, and personal values.

About the author
Sofia Rossi, MD, is a board-certified obstetrician-gynecologist with over 15 years of experience in high-risk pregnancies and reproductive health. She is a clinical professor at a top New York medical school and an attending physician at a university hospital.