The Tyrer-Cuzick Model: A Comprehensive Guide to Breast Cancer Risk Assessment

Key points
- Current Age, Height, and Weight: Used to calculate Body Mass Index (BMI). While premenopausal BMI has a complex or even protective relationship with breast cancer risk, postmenopausal obesity is a well-established independent risk factor due to peripheral aromatization of androgens to estrogens in adipose tissue. Higher BMI after menopause significantly elevates lifetime risk.
- Age at First Menstrual Period (Menarche): An earlier start can slightly increase lifetime risk. Each year of early menarche increases cumulative estrogen exposure before the breast tissue fully matures, providing a longer window of hormonal stimulation that can promote cellular proliferation and potential oncogenic mutations.
- Childbirth History: Includes whether you have given birth and your age at your first live birth. Nulliparity (never having given birth) and first full-term pregnancy after age 30 are associated with elevated risk. Pregnancy induces terminal differentiation of mammary epithelial cells, which confers long-term protection against malignant transformation. Conversely, a transient increase in risk occurs for several years postpartum due to hormonal surges and breast tissue remodeling.
- Menopausal Status: Whether you are pre-menopausal, in menopause, or post-menopausal. The model adjusts risk calculations based on menopausal transition, as the primary sources of sex hormones shift from the ovaries to peripheral adipose tissue and adrenal glands after menopause.
- Hormone Replacement Therapy (HRT): Current or past use is considered. Combined estrogen-progestin therapy, particularly when used for more than five years, has been shown in large randomized trials to modestly increase breast cancer risk by promoting cellular proliferation and potentially delaying apoptosis. The model differentiates between type of HRT, duration of use, and time since cessation.
- Oral Contraceptive Use: While modern low-dose formulations carry minimal absolute risk, prolonged use before age 25 is factored into the algorithm, as younger breast tissue is more hormonally responsive.
The Tyrer-Cuzick model has emerged as a crucial tool in the proactive management of breast health, gaining significant public attention after actress Olivia Munn credited it with the early detection of her breast cancer. This comprehensive statistical model provides a personalized risk assessment, empowering individuals and their doctors to make more informed decisions about screening and prevention.
Breast cancer remains the most commonly diagnosed malignancy among women globally, and early detection is universally recognized as the single most effective strategy for reducing mortality. Historically, screening guidelines relied on age-based, population-wide protocols that assumed an average risk for all women. However, modern oncology and preventive medicine have shifted toward risk-adapted strategies, recognizing that breast cancer susceptibility varies dramatically based on genetics, reproductive history, tissue composition, and environmental exposures. In this paradigm shift, the Tyrer-Cuzick model has become the gold standard for individualized risk stratification, bridging the gap between population-level epidemiology and personalized patient care.
This article synthesizes extensive research and clinical data to offer a complete overview of the Tyrer-Cuzick model, what your score means, its accuracy, and how it compares to other risk assessment tools. By understanding the mechanics, clinical applications, and limitations of this algorithm, patients and healthcare providers can collaboratively design screening and prevention strategies that maximize early detection while minimizing unnecessary interventions.
What is the Tyrer-Cuzick Model?
The Tyrer-Cuzick Model, also known as the IBIS (International Breast Cancer Intervention Study) Risk Evaluation Tool, is a sophisticated algorithm that estimates a woman's likelihood of developing invasive breast cancer over the next 10 years and throughout her lifetime. Unlike simpler models, it integrates a wide range of personal and family health factors to create a more nuanced and individualized risk profile. Developed originally by Professor Jack Cuzick and Professor Jonathan Tyrer in the early 2000s, the model has undergone multiple iterative updates. Version 7 and subsequently Version 8 incorporated mammographic density as a major independent variable, significantly enhancing its predictive power. Version 8 also expanded the genetic component to account for polygenic risk scores and additional susceptibility genes beyond BRCA1/BRCA2, such as CHEK2, PALB2, and ATM.
The mathematical foundation of the Tyrer-Cuzick model relies on complex statistical methods that combine pedigree analysis, population incidence data, and established epidemiological risk factors. It utilizes a modified Claus model framework for familial risk and incorporates age-specific incidence rates for BRCA mutation carriers and non-carriers. By calculating the probability of carrying an underlying high-penetrance gene mutation based on family history patterns, and then applying baseline population risks modified by personal health variables, the algorithm produces a highly individualized risk percentage.
It's essential to understand that the Tyrer-Cuzick model is a risk assessment tool, not a diagnostic test. A high score does not mean you have or will certainly get cancer; rather, it identifies individuals who may benefit from a more personalized and intensive screening strategy. The model calculates absolute risk, which represents the actual percentage chance of developing the disease within a defined timeframe, rather than relative risk, which compares an individual's likelihood to that of the general population. This absolute risk metric is clinically actionable, as it directly aligns with guideline-defined thresholds for supplemental imaging and preventive interventions.
The interface of a Tyrer-Cuzick risk calculator. Source: Volpara Health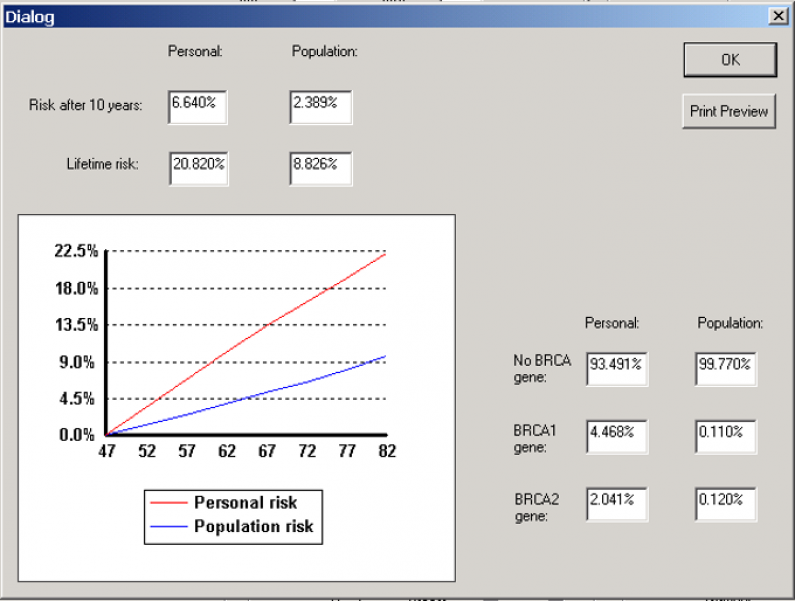
Key Factors Considered in the Tyrer-Cuzick Calculation
The strength of the Tyrer-Cuzick model lies in its comprehensive approach. The calculation incorporates numerous variables to provide a detailed risk score. Each data point contributes a weighted value to the final algorithm, reflecting decades of epidemiological research on breast cancer etiology.
Personal Health and Reproductive History
Reproductive and endocrine factors profoundly influence cumulative lifetime exposure to estrogen and progesterone, which are key drivers in the majority of hormone receptor-positive breast cancers.
- Current Age, Height, and Weight: Used to calculate Body Mass Index (BMI). While premenopausal BMI has a complex or even protective relationship with breast cancer risk, postmenopausal obesity is a well-established independent risk factor due to peripheral aromatization of androgens to estrogens in adipose tissue. Higher BMI after menopause significantly elevates lifetime risk.
- Age at First Menstrual Period (Menarche): An earlier start can slightly increase lifetime risk. Each year of early menarche increases cumulative estrogen exposure before the breast tissue fully matures, providing a longer window of hormonal stimulation that can promote cellular proliferation and potential oncogenic mutations.
- Childbirth History: Includes whether you have given birth and your age at your first live birth. Nulliparity (never having given birth) and first full-term pregnancy after age 30 are associated with elevated risk. Pregnancy induces terminal differentiation of mammary epithelial cells, which confers long-term protection against malignant transformation. Conversely, a transient increase in risk occurs for several years postpartum due to hormonal surges and breast tissue remodeling.
- Menopausal Status: Whether you are pre-menopausal, in menopause, or post-menopausal. The model adjusts risk calculations based on menopausal transition, as the primary sources of sex hormones shift from the ovaries to peripheral adipose tissue and adrenal glands after menopause.
- Hormone Replacement Therapy (HRT): Current or past use is considered. Combined estrogen-progestin therapy, particularly when used for more than five years, has been shown in large randomized trials to modestly increase breast cancer risk by promoting cellular proliferation and potentially delaying apoptosis. The model differentiates between type of HRT, duration of use, and time since cessation.
- Oral Contraceptive Use: While modern low-dose formulations carry minimal absolute risk, prolonged use before age 25 is factored into the algorithm, as younger breast tissue is more hormonally responsive.
Breast Health and Genetic History
Genetic predisposition and prior histological findings are among the most powerful predictors in risk modeling.
- History of Breast Biopsies: The model specifically accounts for findings like atypical hyperplasia or lobular carcinoma in situ (LCIS), which are significant risk factors. Atypical ductal hyperplasia (ADH) and ALH/LCIS are considered high-risk proliferative lesions, conferring a 4 to 5-fold increased risk for subsequent invasive carcinoma. These lesions indicate an unstable epithelial microenvironment prone to malignant progression.
- Personal History of Ovarian Cancer: A personal history increases breast cancer risk, particularly in the context of shared genetic pathways like BRCA mutations. Even in sporadic ovarian cancer cases, shared endocrine and inflammatory pathways can elevate subsequent breast cancer risk.
- BRCA1/BRCA2 Gene Mutations: Known status of these high-risk genes is a critical input. BRCA1 mutations are associated with a 60-75% lifetime risk, predominantly triple-negative breast cancers. BRCA2 mutations carry a 50-70% lifetime risk, typically hormone receptor-positive. The model also integrates testing results for intermediate-penetrance genes when available.
- Family History: The model gathers detailed information on first-degree (mother, sister, daughter) and second-degree (aunt, grandmother) relatives with breast or ovarian cancer. It specifically accounts for age at diagnosis in relatives (premature onset <50 years carries greater weight), bilateral disease, male breast cancer, and multiple affected generations, using pedigree analysis to estimate mutation probability.
- Ashkenazi Inheritance: This ancestry is associated with a higher prevalence of BRCA mutations due to founder effects, specifically the 185delAG and 5382insC mutations in BRCA1 and the 6174delT mutation in BRCA2.
The Critical Role of Breast Density
A pivotal component of the latest versions of the model (v8 and higher) is the inclusion of breast density. Dense breast tissue appears white on a mammogram and is composed of more fibroglandular and stromal tissue relative to adipose tissue. Dense breast tissue is a significant independent risk factor for breast cancer, increasing risk 1.2 to 2 times compared to fatty breasts. Integrating this data, often from a mammogram report (BI-RADS score), has been shown to substantially improve the model's accuracy. Additionally, dense tissue creates a masking effect, where both cancerous and benign lesions appear white, significantly reducing mammographic sensitivity. By incorporating density, the Tyrer-Cuzick model simultaneously addresses biological risk and screening sensitivity, allowing clinicians to recommend supplemental imaging that compensates for the masking effect.
Understanding Your Tyrer-Cuzick Score: What the Numbers Mean
After inputting the data, the model generates a 10-year and a lifetime risk percentage. Healthcare providers use these scores to categorize risk and guide screening recommendations. The transition from a numerical score to a clinical care pathway is where the model proves its greatest utility.
| Risk Category | Lifetime Risk Score | Clinical Guidance |
|---|---|---|
| Average Risk | < 15% | Continued annual screening mammography is typically recommended. |
| Intermediate Risk | 15% - 19.9% | Supplemental screening (e.g., breast MRI or ultrasound) may be considered, especially for women with dense breasts. A discussion with your doctor is advised. |
| High Risk | ≥ 20% | You are considered at high risk. Annual screening mammography and annual breast MRI are often recommended. A referral to a high-risk breast clinic or genetic counselor may also be advised. |
The 20% lifetime risk threshold is a widely accepted benchmark used by organizations like the American College of Radiology (ACR), National Comprehensive Cancer Network (NCCN), and American Cancer Society (ACS) to recommend more intensive screening. However, clinical interpretation extends beyond this binary cutoff.
For women in the average risk category, standard screening typically involves digital breast tomosynthesis (3D mammography) annually or biennially starting at age 40, depending on institutional protocols and patient preference. Shared decision-making is encouraged to weigh the benefits of early detection against the potential harms of false positives and overdiagnosis.
Women classified as intermediate risk often fall into a clinical gray zone that requires careful navigation. Factors pushing toward supplemental imaging include extremely dense breast tissue (BI-RADS C or D), borderline family history, or a 10-year risk that exceeds age-adjusted population averages. In this group, contrast-enhanced spectral mammography (CESM) or abbreviated breast MRI protocols are increasingly utilized as cost-effective adjuncts to traditional mammography.
The high risk category (≥20% lifetime risk) triggers a comprehensive risk management protocol. The ACR and Society of Breast Imaging recommend annual screening breast MRI in addition to mammography for this cohort. MRI is vastly more sensitive than mammography in dense tissue and can detect invasive and in situ malignancies months to years before they become palpable or visible on standard imaging. Many high-risk screening programs stagger MRI and mammography six months apart to provide semi-annual surveillance. Furthermore, women in this category are strongly encouraged to undergo formal genetic counseling and testing if they haven't already, and to discuss chemoprevention or surgical risk-reduction strategies with a multidisciplinary team.
How Accurate and Reliable is the Tyrer-Cuzick Model?
The Tyrer-Cuzick model is widely regarded as one of the most robust breast cancer risk assessment tools available. Its dominance in clinical guidelines stems from rigorous external validation, continuous algorithmic refinement, and alignment with real-world clinical outcomes.
Strengths and Validated Performance
- Comprehensive Nature: Its inclusion of numerous variables, especially detailed family history and breast density, makes it more accurate for many women compared to simpler models. By simultaneously evaluating hereditary susceptibility and environmental/hormonal exposures, it avoids the oversimplification that plagues less sophisticated calculators.
- Long-Term Accuracy: A major study published in JAMA Oncology followed women for nearly 19 years and found the model was a well-calibrated and accurate predictor of long-term risk. The model demonstrated excellent calibration, meaning the predicted number of cases closely matched the observed number across all risk deciles. Its Area Under the Curve (AUC) or C-statistic consistently ranges between 0.65 and 0.70, which is considered strong for epidemiological prediction models.
- Improved Discrimination: By incorporating more risk factors, it is better at distinguishing between women who will develop breast cancer and those who will not. The integration of mammographic density in v8 specifically boosted discrimination metrics, particularly in younger women and those with dense breasts where older models consistently underperformed.
Important Limitations and Considerations
Despite its strengths, the model has limitations that users and clinicians must acknowledge. Risk modeling is an evolving science, and no algorithm can capture the infinite complexity of human biology and environmental interactions.
- It Is Not a Crystal Ball: The score is a statistical probability, not a definitive prediction. Many women with a high-risk score will never develop breast cancer, and some with a low-risk score will. Risk estimates represent population-level probabilities applied to an individual, and stochastic (random) biological events still play a major role in carcinogenesis.
- Lifestyle Factors: The model does not directly incorporate lifestyle factors like diet, exercise, and alcohol consumption, which also influence risk. Chronic alcohol intake (more than 1 drink/day), sedentary behavior, and diets high in processed foods contribute to systemic inflammation, insulin resistance, and altered hormone metabolism, all of which modulate breast cancer risk independently.
- Data Quality Dependency: The accuracy of the output is entirely dependent on the accuracy of the input. Incomplete family histories, forgotten medication usage, or inaccurate self-reporting of menopausal status can skew results. Clinicians often spend significant time verifying pedigree data before finalizing the risk calculation.
- Dynamic Nature of Risk: A Tyrer-Cuzick score is not a permanent value. It changes over time as a woman ages, transitions through menopause, undergoes new biopsies, modifies HRT usage, or receives updated genetic testing results. Regular recalculation every 1-2 years or following any major health event is standard clinical practice.
Performance in Diverse Populations: A Critical Gap
One of the most significant limitations is that the model was developed and validated primarily in cohorts of White women. Emerging research indicates its performance may vary across different racial and ethnic groups. Healthcare systems must approach risk stratification with cultural competence and awareness of epidemiological disparities.
- Underestimation in Black Women: Studies suggest the model may underestimate risk in Black women. Black women experience disproportionately higher rates of aggressive, early-onset, and triple-negative breast cancer. Biological differences in tumor biology, alongside socioeconomic and environmental stressors not captured by the model, contribute to this discrepancy.
- Overestimation in Hispanic Women: Conversely, some research indicates a potential overestimation of risk for Hispanic women, particularly those of Mexican descent, who generally exhibit lower baseline incidence rates for breast cancer but higher mortality when diagnosed, often due to later-stage presentation.
This highlights an urgent need for further validation studies in large, diverse populations to refine the model and ensure equitable and accurate risk assessment for all women. Ongoing research initiatives are working to recalibrate the algorithm using multi-ethnic cohorts and incorporate social determinants of health, neighborhood environment, and access to care metrics to close this predictive gap.
Tyrer-Cuzick vs. Other Breast Cancer Risk Models
Clinicians have several risk models to choose from, each with specific strengths. The selection of a particular model depends on clinical context, data availability, and the specific clinical question being asked (e.g., screening eligibility vs. chemoprevention eligibility vs. genetic testing thresholds).
!A table comparing the variables used in different breast cancer risk models, including Gail, Claus, and Tyrer-Cuzick.
Comparison of variables used in different risk models. Source: ResearchGate
| Model | Primary Use Case | Key Strengths | Key Weaknesses |
|---|---|---|---|
| Tyrer-Cuzick | Personalized screening decisions (e.g., MRI eligibility). | Highly comprehensive, includes breast density, detailed family history, and genetic factors. | More complex to use; less validated in diverse populations. |
| Gail Model | Determining eligibility for chemoprevention (risk-reducing drugs). | Simple, fast, and widely validated for its specific purpose. | Considers limited family history; does not include breast density; may underestimate risk. |
| BRCAPRO | Estimating the probability of carrying a BRCA1/BRCA2 mutation. | Specialized and accurate for predicting genetic mutation carriers. | Not a comprehensive risk model for the general population. |
A study comparing models in women with a family history of cancer found that the Gail model tended to underestimate risk, concluding the Tyrer-Cuzick model was more appropriate for that population.
The Gail Model (BCRAT) is heavily utilized by primary care physicians because of its simplicity, requiring only age, reproductive history, and limited first-degree family history. It was originally designed to identify candidates for chemoprevention trials and is FDA-cleared for determining eligibility for tamoxifen or raloxifene use in women over 35. However, it excludes second-degree relatives, paternal family history, age of onset in relatives, breast density, and ovarian cancer history. Consequently, in women with strong family histories, Gail consistently underestimates true risk, making Tyrer-Cuzick the preferred choice for screening pathway decisions.
The Claus Model focuses exclusively on family history and genetic inheritance patterns, calculating risk based on pedigree analysis without incorporating personal reproductive or lifestyle factors. It is largely considered outdated for standalone clinical use because it ignores well-established non-genetic risk modifiers.
The BRCAPRO model, part of the BayesMendel suite, is highly specialized. It uses Bayesian probability to predict the likelihood that a patient carries a BRCA1 or BRCA2 mutation based on detailed family cancer histories. While excellent for pre-test genetic counseling, it does not predict overall breast cancer risk in non-carriers or account for density, HRT, or biopsy history, limiting its scope to genetic risk evaluation rather than comprehensive screening planning.
In modern clinical practice, Tyrer-Cuzick has effectively become the default for comprehensive risk assessment, particularly within breast imaging centers and oncology networks. Major guidelines, including the NCCN Breast Cancer Screening and Diagnosis guidelines and the ACR Appropriateness Criteria, explicitly reference the 20% lifetime risk threshold calculated by Tyrer-Cuzick (or models with similar comprehensiveness) to justify annual MRI screening.
From Score to Action: Clinical Implementation
A Tyrer-Cuzick score is most valuable when used in a clinical setting to create an actionable plan. Raw data without a management pathway is clinically inert; therefore, healthcare systems are increasingly embedding risk calculation into standardized clinical workflows.
- Risk Calculation: The score is often calculated at the time of a mammogram. Radiologic technologists or patient navigators collect the necessary questionnaire data before imaging. Increasingly, AI-driven natural language processing extracts relevant data directly from electronic chart notes to auto-populate the calculator.
- Personalized Recommendations: Based on the score, a radiologist or referring physician will recommend a screening plan. For average-risk patients, this means standard annual tomosynthesis. For high-risk patients, it triggers MRI scheduling, insurance pre-authorization letters citing specific ACR guidelines, and automated referrals to genetics or high-risk clinics.
- Patient-Doctor Discussion: The score facilitates a crucial conversation about personal risk, the benefits and limitations of additional screening, and potential risk-reduction strategies. Shared decision-making ensures the patient understands why additional imaging is recommended, what to expect during an MRI (including contrast administration, claustrophobia management, and acoustic noise), and how to interpret potential false positives.
Risk-Reduction Strategies for High-Risk Individuals
Beyond enhanced screening, a high Tyrer-Cuzick score opens the door to proactive risk reduction. Clinical guidelines outline three primary tiers of intervention:
- Lifestyle and Behavioral Modification: Maintaining a healthy BMI, engaging in at least 150 minutes of moderate-intensity aerobic activity weekly, limiting alcohol to ≤1 drink per day, and avoiding combined HRT when possible can reduce breast cancer risk by 15-25%.
- Chemoprevention: For women at high risk, risk-reducing medications have proven efficacy. Selective Estrogen Receptor Modulators (SERMs) like tamoxifen (for pre- and post-menopausal) and raloxifene (post-menopausal only) reduce estrogen receptor-positive breast cancer risk by approximately 50%. Aromatase inhibitors like exemestane and anastrozole are also FDA-approved for post-menopausal high-risk women and demonstrate similar or superior efficacy with different side effect profiles. Despite their proven benefit, uptake remains low due to concerns about menopausal symptoms, thrombosis risk, and rare endometrial changes, necessitating thorough counseling.
- Risk-Reducing Surgery: For women with confirmed BRCA mutations or exceptionally high lifetime risks (>30-40%) with strong family anxiety, prophylactic bilateral mastectomy can reduce breast cancer risk by 90% or more. Bilateral salpingo-oophorectomy is also recommended for BRCA carriers, typically between ages 35-40, as it drastically reduces ovarian cancer risk and further lowers breast cancer risk by eliminating ovarian hormone production.
Innovations are also making this process easier. Companies are integrating Tyrer-Cuzick calculators directly into Electronic Health Record (EHR) systems, allowing clinicians to access these powerful insights seamlessly within their workflow. Automated clinical decision support (CDS) tools flag patients exceeding risk thresholds, generate standardized referral templates, and track longitudinal risk changes across care episodes, reducing clinician cognitive load and improving adherence to screening guidelines.
How Can You Get Your Tyrer-Cuzick Score?
Many imaging centers and breast health clinics now offer Tyrer-Cuzick risk assessment as part of their standard mammography services. The widespread adoption of the Breast Cancer Risk Assessment (BCRA) component in mammography reporting, driven by FDA-mandated density reporting laws and ACR recommendations, has normalized risk stratification as a standard of care. Additionally, several online calculators are available for public use. While these can be informative, it is vital to discuss the results with a healthcare provider for accurate interpretation and clinical guidance. Self-calculated scores lack the clinical context, pedigree verification, and histological review that a trained provider offers.
- IBIS Risk Evaluation Tool (official): ems-trials.org/riskevaluator/
- Ikonopedia Online Calculator: ibis.ikonopedia.com/
When utilizing online tools, patients should prepare by gathering detailed family cancer histories, including ages at diagnosis, types of cancer, and survival status. Patients should also locate prior mammogram reports to accurately input their BI-RADS density category and recall any past biopsies or gynecological surgeries. If you receive a high-risk score online, do not panic; schedule an appointment with your primary care physician, gynecologist, or a breast specialist to verify the calculation, review your personal medical history, and develop a coordinated care plan. Conversely, if you have a strong family history but a low calculated score, still share your concerns with a provider, as rare syndromes or non-standard inheritance patterns may warrant genetic testing regardless of the model's output.
Insurance coverage for supplemental screening has expanded significantly. Under the Affordable Care Act (ACA), screening mammograms are covered without cost-sharing, and many states have passed supplemental density screening mandates. Furthermore, as more insurers recognize the clinical validity of Tyrer-Cuzick-based risk stratification, they increasingly cover breast MRI for high-risk patients without excessive prior authorization hurdles. Patients should work with their provider's billing department to verify coverage, and high-risk clinics often employ financial navigators to assist with appeals and alternative payment programs.
Conclusion: An Empowering Tool for Proactive Breast Health
The Tyrer-Cuzick model is a powerful and comprehensive tool that has transformed breast cancer risk assessment. By providing a personalized, data-driven estimate of risk, it enables a shift from one-size-fits-all screening to a more tailored and effective strategy. The integration of genetic, familial, hormonal, and morphological (density) data represents the pinnacle of current preventive oncology, allowing clinicians to move from reactive diagnosis to proactive surveillance and risk modification.
While it is not perfect and requires further validation in diverse populations, it represents a significant step forward in empowering individuals to take proactive control of their breast health. As research advances, we can expect future iterations to incorporate polygenic risk scores, microbiome data, advanced proteomic biomarkers, and artificial learning algorithms to further refine predictive accuracy. The ultimate goal is not just to find cancer earlier, but to stratify risk so precisely that we can offer the right intervention, to the right patient, at the right time. Always remember to discuss your risk score and screening options with your doctor to create the best plan for you. Your health journey is unique, and risk assessment is the compass that helps you and your medical team navigate it with confidence and precision.
References
- Ambry Genetics. (2025). Ambry Genetics Announces Improved EHR-Integrated Breast Cancer Risk Assessment Solution. Business Wire. Link
- Brentnall, A. R., & Cuzick, J. (2018). Use of the Tyrer-Cuzick model for breast cancer risk prediction. Breast Cancer Management. Link
- MagView. Comparison of the Tyrer-Cuzick vs Gail Risk Assessment. Link
- National Breast Cancer Foundation. (2025). The Tyrer-Cuzick Model: Understanding the Assessment. Link
- OnlyMyHealth. (2025). What Is The Tyrer-Cuzick Risk Model. Link
- Vianna, F. S. L., et al. (2019). Performance of the Gail and Tyrer-Cuzick breast cancer risk models. Mastology. Link
Frequently Asked Questions
How often should I recalculate my Tyrer-Cuzick risk score?
Your Tyrer-Cuzick score should be recalculated whenever a significant health event occurs or at regular intervals during routine breast care. Key triggers for recalculation include aging into a new decade (as risk naturally increases with age), transitioning to menopause, starting or stopping hormone replacement therapy, experiencing a new breast biopsy (especially if atypical hyperplasia is found), or receiving updated genetic testing results. If none of these events occur, most breast specialists recommend repeating the assessment every 1 to 2 years alongside your routine mammography to ensure your screening plan remains appropriately calibrated to your current risk profile.
Does a high Tyrer-Cuzick score mean I need to have a mastectomy?
Absolutely not. A high score indicates elevated statistical risk, not a certainty of disease, and prophylactic mastectomy is just one of several management options. The vast majority of women with a high Tyrer-Cuzick score manage their risk through enhanced surveillance (annual mammography plus breast MRI) and lifestyle modifications. Chemoprevention medications like tamoxifen, raloxifene, or aromatase inhibitors are highly effective, non-surgical alternatives that can reduce risk by approximately 50%. Risk-reducing surgery is typically reserved for individuals with confirmed high-penetrance genetic mutations (like BRCA1/2), those with extremely strong family histories, or patients who express profound anxiety about cancer despite understanding the statistics. The decision is deeply personal and should be made collaboratively with a multidisciplinary team including surgeons, oncologists, and genetic counselors.
Can I use the Tyrer-Cuzick model if I have a strong family history of ovarian or other cancers?
Yes, the Tyrer-Cuzick model explicitly accounts for family histories of ovarian cancer, which is a crucial feature since BRCA mutations and other hereditary syndromes increase the risk for both breast and ovarian malignancies. When entering data, you should include all first- and second-degree relatives with ovarian cancer, noting their age at diagnosis. The algorithm uses this information to calculate the probability of an underlying genetic mutation, which subsequently adjusts your breast cancer risk upward. If you have a family history of male breast cancer, pancreatic cancer, or prostate cancer at young ages, be sure to share this with your provider as well, as it may prompt referral for comprehensive multi-gene panel testing beyond standard breast cancer risk assessment.
Is supplemental breast MRI covered by insurance if I meet the high-risk threshold?
In most cases, yes. The Affordable Care Act and numerous state-mandated density and high-risk screening laws require insurance plans to cover screening mammograms without cost-sharing, and an increasing number of states have passed laws specifically mandating coverage for supplemental screening, including breast MRI, when medically indicated. When your Tyrer-Cuzick score is 20% or greater, you meet the widely accepted clinical criteria established by the American College of Radiology and the American Cancer Society. Most commercial insurers and Medicare now recognize this threshold as justification for annual screening MRI. However, coverage details vary by plan and region, so your provider's office will typically submit pre-authorization documentation citing your calculated risk score, density category, and clinical guidelines to secure approval before scheduling.
Why might my online calculator score differ from the score calculated at my imaging center?
Discrepancies between self-administered online calculators and clinical systems are common and usually stem from differences in data completeness, model version, and professional interpretation. Clinical centers use the most current validated software version (often v8 or higher), which automatically incorporates mammographic breast density from your radiology report—a variable many online users either omit or estimate incorrectly. Additionally, trained staff verify family pedigrees, clarify ambiguous medical history (such as distinguishing between a benign fibroadenoma biopsy and atypical hyperplasia), and ensure all hormonal and reproductive variables are accurately coded. Self-reported age at menarche, exact dates of medication use, and precise family cancer ages can also vary. Always defer to the clinically verified score, as it serves as the legal and medical basis for your personalized screening recommendations.

About the author
Sofia Rossi, MD, is a board-certified obstetrician-gynecologist with over 15 years of experience in high-risk pregnancies and reproductive health. She is a clinical professor at a top New York medical school and an attending physician at a university hospital.